
Groups 3-12 comprise the transition metals. This photo by Alchemist-hp is licensed under CC BY-SA 2.0 DE 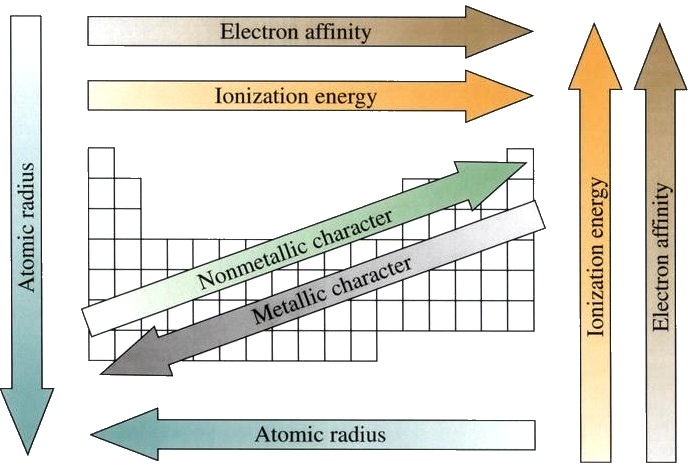
Spectrum = gas discharge tubes: the noble gases: helium He, neon Ne, argon Ar, krypton Kr, xenon Xe. This is why noble gases (except Rn) are commonly used in neon signs. They are also colorless, although ionizing them may cause these gases to emit bright colors. As a result, noble gases are chemically inert. Aside from helium, all members of the group have 8 electrons in their valence shell. Group 18 ( noble gases) have filled p subshells (except He). Furthermore, these two groups are sometimes referred to as the s block elements. Together with the alkali metals, groups 1 and 2 (except H and Be) form strong inorganic bases when bonded with hydroxide ions. Elements under this family have two electrons in their valence shell which they readily lose to form cations with 2 charges. Group 2, on the other hand, is known as alkaline earth metals. As a result, these elements readily lose one electron to form ions with a 1 charge. Elements under this family are highly reactive and possess a single electron in their outermost shell. All the representative elements have their highest s or p orbital incompletely filled.īelonging to the representative elements, group 1 is known as the alkali metals. Although the law was able to group a few elements with similar properties successfully, it was generally considered unsuccessful.Įlements in groups 1, 2, and 3-17 are the representative elements or the main group elements. Newland observed that elements are arranged based on their atomic weights, elemental properties seem to repeat every eight elements. Atomic masses of some Dobereiner’s triad.ĭobereiner’s model was followed by John Newland’s law of octaves in 1864. Element Symbol Atomic Mass Lithium Li 6.9 Sodium Na 23.0 Potassium K 39.0 Chlorine Cl 35.4 Bromine Br 79.9 Iodine I 126.9 Sulfur S 32.0 Selenium Se 79.0 Tellurium Te 128.0 Table 1. Table 1 enumerates some of Dobereiner’s triads. Unfortunately, it fails to apply to most of the elements known during Dobereiner’s time, proving the model severely lacking. The model states that the atomic weight of the middle element in a set of three elements (he called triad) is approximately equal to the arithmetic mean of the two extreme elements in the set.ĭobereiner was able to identify several triads of elements with the same properties which adhere to this model. The development of the periodic table as we know it today started in 1829 when Johann Dobereiner proposed the model of triads or the law of triads in some references. 2. Answer Key The Periodic Table of Elements.Groups and Families in the Periodic Table.the pair of dots represent valence electrons that are not involved in bonding, called lone pair of electrons.When you hear the word “Chemistry,” what comes to mind? There may be a lot of things, but one of those is probably the periodic table of the elements! In this chapter, we will talk about how elements are arranged in the modern periodic table and what important properties and trends we can obtain from this arrangement.Each line in these molecules represents a bonding electron pair, and.Carbon, nitrogen, oxygen, and fluorine with 4, 3, 2, and 1 unpaired dot can make 4, 3, 2, and 1 bond, e.g., in the following molecules:, ,, and. A bond is formed by sharing unpaired valence electrons. A bond is represented by a line between the bonded atoms. Generally, each unpaired dot can make one bond.įor example, a hydrogen atom with one unaired dot can make one bond as in H-H. The electron dots in the Lewis structure are a convenient way to determine how many bonds an atom of an element can make. \): Lewis symbols or electron-dot symbols of the first twenty elements in the periodic table. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
